1. What is flotation separation?
Flotation separation mainly refers to froth flotation, which is based on the difference in physical and chemical properties of the surface of minerals, using the buoyancy of bubbles in the pulp to achieve separation.

The flotation separation process mainly includes: (1) Grind the ore finely by the ball mill or rod mill to dissociate useful minerals from gangue minerals. (2) Separate the ore by the flotation machine. Adjust the ore pulp and add flotation reagents. (3) Do the flotation processing. (4) Filter, concentrate or dry the product.
Click to know more about the flotation plant.
2. Flotation process and types
- Positive flotation: The foams that float on the surface of the pulp are useful minerals, while the gangue minerals remain in the pulp.
- Reverse flotation: The foams that float on the surface of the pulp are the gangue minerals, while the useful minerals remain in the pulp.
- Differential flotation: It separates the useful minerals to be a single concentrate in turn.
- Bulk flotation: It separates the useful minerals together and then separates each of them in turn.
3. Significance of flotation separation
- It is the most widely used and most effective beneficiation method for the separation of fine-grained and particulate minerals.
- It occupies an important position in various beneficiation methods, with a wide range of applications. It can process non-ferrous metal, non-metal and ferrous metal minerals.
- Compared with gravity separation and magnetic separation, flotation separation is more efficient to beneficiate ores, especially the fine-grained disseminated ores. Low-grade raw ore can be separated into a high-grade concentrate. It solves the problem of the recovery of useful components from fine-grained mineral particles, thereby having a high recovery rate.
4. How to judge mineral floatability?
The criterion for judging mineral floatability is mineral wettability.
Minerals that are not easily wetted by water (hydrophobic) (graphite, chalcopyrite, gold ore, etc.) are considered buoyant or floatable. Otherwise, minerals (such as quartz) are difficult to float.
Wetting is a basic phenomenon that occurs at the solid-liquid interface. When a drop of liquid is placed on the mineral surface, the drop will spread out on the surface. This phenomenon is called the wetting of the surface of the solid phase by the liquid phase.

If water can spread on the mineral surface, the mineral is hydrophilic. Otherwise, it is hydrophobic. For example, water can spread out on the quartz surface, so quartz is hydrophilic. The paraffin is hydrophobic.
The speed of wetting is very important. As various minerals have differences in wettability and speed, mineral particles can selectively attach to air bubbles. Certain flotation reagents can change wettability. For example, that scheelite (CaWO4) adsorbs oleic acid can change the mineral from hydrophilic to hydrophobic.
5. Surface electrical property and floatability of minerals
The preferential adsorption, preferential dissociation, and lattice substitution of ions can charge the mineral surface in the ore pulp, which can affect the floatability of some minerals.
- For the Oxide minerals and silicates, such as goethite, corundum, and quartz, the surface electrical property will greatly affect the floatability. It lies in that these minerals will have electrostatic adsorption with collectors. The symbols of surface electrical property are related to the pH value of the ore pulp.
- For the sulfide minerals, such as copper, lead, and zinc, collected with xanthate, the surface electrical property has little or no effect on the floatability. That's because the action of xanthate with minerals mainly depends on the chemical affinity rather than the electrostatic force.
- In addition, for the non-sulfide minerals collected by oil collectors, the surface electrical property does not affect the floatability.
6. Adsorption forms of reagents on the mineral surface
- Molecular adsorption: The reagent is adsorbed on the mineral surface in the form of molecules.
- Ion adsorption: The reagent is adsorbed on the mineral surface in the form of ions.
- Ion exchange adsorption: The reagent is absorbed on the mineral surface when the certain ion in the ore pulp has an equivalent exchange with another ion on mineral surface that has the same charge sign.
- Electrical adsorption: The reagent is absorbed on the mineral surface by the electrostatic force.
- Semi-micelle adsorption: When the mass fraction of collectors of a long hydrocarbon chain in the slurry is high, their ions or molecules are adsorbed on the mineral surface. Under the Van der Waals force, the non-polar group will associate with each other.
- Specific adsorption: The mineral surface has a special affinity for a certain component in the slurry to produce adsorption.
7. Effect of oxidation of mineral surface on floatability
Minerals are oxidized by air during storage, transportation, crushing, and flotation. The oxidation of minerals, especially metal sulfide ore, can greatly affect the flotation.
Research shows that some sulfide minerals have better floatability after being slightly oxidized. However, if they are over-oxidized, the floatability will decrease.

Air has certain effects on mineral floatability
- When the mineral is crushed and exposed to the fresh surface, it will be hydrated when it encounters water, and it will be hydrophilic. However, when the gas is adsorbed to the mineral surface, it can weaken the hydration, resulting in the initial hydrophobicity of the surface.
- The use of gas and mineral surfaces is selective. Oxygen has a greater impact on the mineral surface.
- The role of oxygen is beneficial to the hydrophobicity of sulfide ore. However, if the action time is too long, the mineral surface will become hydrophilic. When the gas adsorption conditions are suitable, the mineral surface will be hydrophobic and can be floated even without flotation reagents (such as dry coal powders). And galena can only float up after the initial action of oxygen.
How to control the minerals' oxidation degree and adjust the floatability?
- Adjust the speed of slurry agitation and control the flotation time. It is proven that the speed of air agitation and duration are important for controlling the oxidation degree of minerals.
- Adjust the aeration of the pulp tank and flotation machine.
- Adjust the pH value of the ore pulp.
- Add the oxidant or reductant to promote or inhibit the oxidation of the mineral surface.
8. Selective flocculation
Selective flocculation can effectively separate fine-grained minerals.

Its principle is to add the flocculant into the suspension that contains two or more compositions. The flocculant is selectively adsorbed on the surface of a certain mineral composition to promote its flocculation and sedimentation. The remaining compositions still maintain a stable dispersion state to achieve the mineral separation.
Selective flocculation can be divided into four steps:
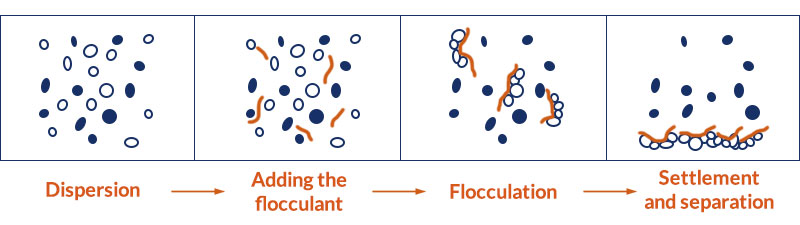
Step 1. Add the dispersant
The dispersant, such as water glass, can fully disperse the useful mineral compositions in the ore pulp to make them have no adhesion and connection with each other, thereby creating a better condition for the selective flocculation.
Step 2. Add the flocculant
The flocculant is selectively adsorbed on the surface of target minerals and meanwhile connects with other mineral particles to form larger, loose, and porous flocs, which settle in the pulp.
Step 3. Stir the ore pulp
When stirring the ore pulp, please pay attention to the following points. Stirring is generally conducted in different stages with quick stirring first to fully disperse the reagents and then full stirring to facilitate the selective flocculation of minerals. The ore pulp should be diluted to facilitate the cleanness of flocculant and reduce the adhesion of other minerals. The flocs should not be too large in case the impurities are entrained.
Step 4. Settlement and separation
When the flocs reach a certain size, settlement and separation shall be carried out. Pay attention to the time and speed of settlement.
The speed of settlement must be slow to prevent impurities from being carried into the sediment. Sometimes the weak water flow can be used to release the impurities from the flocs.
The time of settlement should be appropriate. If the time is too short, the product will be purer, but will affect the recovery rate. If the time is too long, the impurities will also settle to affect the mineral quality. Besides, if the first flocculation fails to meet the product requirements, the flocculation process can be carried out again.
9. Three-phase system of flotation separation
The three-phase system of flotation includes the solid phase, liquid phase, and gas phase.
The solid phase refers to the finely ground mineral particles. The liquid phase refers to the water and solution. The gas phase refers to the dispersed bubbles.
Characteristics of the solid phase
- There are often several useful minerals and gangues associated together, with so many minerals' types and compositions. In terms of shape, the ore particles are square, columnar, flake, and irregular angular.
- It has a wide particle size range. The biggest particle size can reach 0.8 to 1 mm, while the smallest particle size is below 5 mm.
- It has quantities of ore particles with a large surface area. Each liter of the ore pulp can have tens of millions or even nearly 100 million fine particles, thus the surface area of the solid phase is large.
Characteristics of the liquid phase
- Water can interact with the solid and gas phases in the ore pulp, as the ore often contains various soluble salts that can be dissolved in water.
- Industrial natural water often contains some soluble salts.
- Oxygen, nitrogen and carbon dioxide are dissolved in the water. Therefore, the liquid phase is not simply water but is essentially a solution, which can greatly affect the flotation.
Characteristics of the gas phase
- The air appears as the finely dispersed bubbles in the ore pulp, which can carry the mineral particles to float.
- The air can be repeatedly dissolved and precipitated in the pulp.
- The oxygen in the air can greatly affect mineral floatability.
10. How to increase the flotation speed?
The flotation speed refers to the flotation time when a certain recovery rate or unit production capacity of the flotation machine is reached. Thus, by improving the recovery rate or shortening the flotation time, the flotation speed can be increased.
Measures should be taken to increase the flotation speed:- 1Add the appropriate amount of reagents, especially the amount of foaming agent.
- 2Within an appropriate range, increase the speed of the impeller of the flotation machine and reduce the depth of the groove to reduce the gap between the impeller and cover.
- 3Make the ore pulp pass through the flotation tank as soon as possible. The series tank is faster than the parallel tank.
- 4The selected tank must be in an appropriate size.
- 5The pulp density should be appropriate. Thicker pulp is beneficial to the flotation process, but too thick will affect the concentrate quality and the recovery rate.
11. Flotation reagents
Using flotation reagents correctly means choosing the right reagent system before the flotation process.
The flotation reagent system refers to a set of rules regulating the type, dosage, dosing location, and dosing method of flotation reagents.
Click to know more about flotation reagents.

Reagents types
The flotation reagents mainly include foaming agents, collectors, and regulators.
- Foaming agents aim to produce a stable foam layer that can float minerals. Foaming agents include turpentine, cresol, alcohol, etc.
- Collectors aim to collect the target minerals by enhancing the hydrophobicity to make the mineral particles adhere to the air bubbles. The common collectors are areoflot, xanthate, diphenylthiourea, fatty acid, fatty amine, and mineral oil.
- Regulators aim to change the mineral properties, adjust the interaction between minerals and collectors, and adjust the pulp properties. The common regulators include pH regulators (sodium carbonate, sulfuric acid, sulfur dioxide, etc.), activators (copper sulfate, sodium sulfide, etc.), and inhibitors (lime, yellow prussiate of soda, sodium sulfide, etc.).
Xanthate is the collector of sulfide ore with a strong collecting ability. Its chemical name is hydrocarbyl dithiocarbonate, including ethyl xanthate, butyl xanthate, amyl xanthate, etc. It is a yellow crystalline powder with an irritating effect and it should be stored in a cool and dry place.
Dosage of flotation reagents
The reagent dosage should be appropriate.
Insufficient or excessive dosage leads to a low recovery rate and affects the concentrate quality. Excessive dosage also increases the beneficiation cost.
Preparation of flotation reagents
Dilute the solid reagents into a liquid for easy addition.
The preparation of reagents varies with their properties, function, and ways of adding them. The same reagent may differ in the quantity of use and effect due to different preparation ways.

Reagents (such as xanthate, aerofloat, sodium carbonate, and copper sulfate) with poor water solubility should be prepared into water solutions with concentrations ranging from 2% to 10%.
Insoluble reagents like amine collectors should be dissolved in solvents first, and then prepared into an aqueous solution. Terpenic oil, aerofloat 31, and oleic acid can be directly added.
For freely soluble reagents with large quantities to use, the concentration ranges from 10 to 20%. For example, sodium sulfide is prepared into a 15% water solution. Reagents that are insoluble in water can be dissolved using an organic solvent to be prepared into a low concentration solution.
How to add flotation reagents?
The order of adding flotation reagents:- For the flotation of raw ore: pH regulators, depressants or activators, foaming agents, and collectors.
- For the flotation of depressed minerals: activators, foaming agents, and collectors.
Flotation reagents can be added simultaneously or separately.
- Stable water-soluble reagents that are not easily carried away by foam can be added to all reagents at one time before pre-concentration.
- Reagents that are easily carried away by foam or react with fines and soluble salts are added in stages.