Nickel is a hard, silvery-white metal that is a staple of our daily lives and can be found in everything from the coins to batteries to stainless steel.
Nickel, much-loved today, was initially discovered with miners loathing it.

Nickel: the only metal named after the devil
Did you know that nickel takes its name from the German word "Kupfernickel"?
In the 1600s, a dark red ore, often with a green coating, was a source of irritation for copper miners in Saxony, Germany. They tried to extract copper from it but failed. Those who were tasked with processing the mysterious ore began to show signs of poisoning. Many began to experience intense stomach pains, which led to vomiting and diarrhea. Others got vertigo, resulting in accidents and falls. Some who worked the smelters even became delirious, went into shock or died.
The miners concluded the ore was not copper but something disguised as such. Convinced that the deposit was the mischievous work of the devil. They named it Kupfernickel, meaning “Old Nick's copper.” Old Nick was the name of an evil sprite in Saxon mythology, and was also used as a colloquial name for the devil.

Between 1751 and 1754, the Swedish chemist Axel Cronstedt conducted a number of experiments to determine the true nature of kupfernickel. We now know that kupfernickel is nickel arsenide or niccolite. Nickel arsenide is toxic, which explains why miners were poisoned.
After finding that its chemical reactions were not what he would have expected from a copper compound, he heated kupfernickel with charcoal to produce a hard, white metal, whose color alone suggested it could not be copper. Its properties, including its magnetism, led him to conclude that he had isolated a new metallic element.
Cronstedt named the new element nickel, after the kupfernickel from which he had isolated it.
Nickel properties
Nickel is one of the most versatile metals, from the utensils we use in our kitchens to the bridges that connect our cities and the batteries of electric vehicles that will help to enable a low-carbon future. The secret is its outstanding physical and chemical properties.
-

HIGH MELTING POINT 1453°C
-

RESISTS CORROSIONAND OXIDATION
-

HIGHLY DUCTILE
-

ALLOYS READILY
-

MAGNETIC AT ROOM TEMPERATURE
-

CAN BE DEPOSITED BY ELECTROPLATING
-

HAS CATALYTIC PROPERTIES
-

CAN BE FULLY RECYCLED
Source: nickelinstitute
The properties of nickel metal also answer frequently asked questions about it.
Is nickel alloy corrosion resistant?
Yes. But keep in mind that nickel-plated steel is corrosion-resistant, not corrosion-proof.
Nickel is the most corrosion-resistant metal known. Pure Nickel can withstand high temperatures, a variety of chemicals, excessive humidity, and other extreme conditions.
Being highly resistant to corrosion, Nickel is used in making an extensive number of alloys such as wrought nickel, nickel-iron, nickel-chromium, and nickel-copper alloys, etc.
However, we must understand that Nickel is corrosion-resistant and not corrosion-proof. Therefore, when exposed to extreme conditions for a long duration, Nickel may also undergo corrosion forming a nickel oxide layer.
Is nickel magnetic?
Nickel is one of only four metals that are ferromagnetic at room temperature, meaning they are attracted to magnets and are magnetic themselves. The others are iron, cobalt and gadolinium. Nickel is less magnetic than iron or cobalt.
Alnico magnets — a combination of aluminum (Al), nickel (Ni) and cobalt (Co) — are very strong permanent magnets that retain their magnetism even when heated until they glow red.
What is nickel plating?
Nickel plating is a technique of depositing a layer of nickel on another material. The purpose is to change the physical properties of an object, ie, its conductivity, hardness, adhesion, corrosion and erosion resistance, durability, performance and appearance.
There are two types of nickel plating:
Nickel electroplating: In this case, the nickel layer is deposited by an electrolytic process.
Electroless nickel plating: In this case, an autocatalytic reaction is used to do the same thing.
Is nickel recyclable?
Like many other metals, nickel is fully recyclable. It can be recycled again and again without loss of quality.
Nickel is one of the world's most recycled materials. About 68% of nickel is reclaimed from consumer products that are recycled. Common recycled sources of nickel are primarily from stainless steel and old batteries.
Nickel recycling not only promotes a circular economy but also alleviates nickel resource shortages caused by growing EV market.
Nickel demand spurred by the growth of the EV market
Nickel is the main cathode material for lithium-ion batteries. Nickel in batteries helps provide higher energy density and greater storage capacity at a lower cost.
Although electric vehicles (EVs) currently account for a relatively small proportion of global automobile stock, their market share is increasing and is forecast to continue to grow rapidly in the coming years. Some predictions suggest they will make up more than 10% of vehicles by 2025, most of which will be powered by nickel-containing Li-ion batteries.
From only 7% of the total market in 2021, Wood Mackenzie expects battery use to grow to 40% of nickel consumption by 2040. That will push nickel demand to double in size to six million tonnes per year.
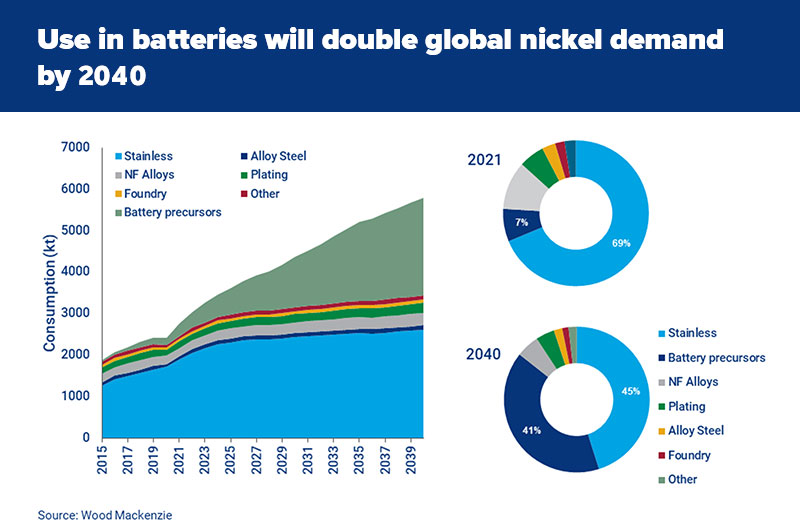
Class 1 nickel needed
Battery makers require Class 1 nickel to produce nickel sulfate – the compound used in lithium-ion battery cathodes. Class I nickel has a nickel content of 99.98% or more and is usually found in nickel sulfide deposits. Unfortunately, large, high quality nickel sulfide deposits are quickly becoming depleted and increasing difficult to find new ones.
While looking for new nickel sulfide ore, we should step up the utilization of nickel laterite resources.
There are two main classifications of laterite ore: limonite and saprolite. Limonitic laterites are easy to mine, but are of lower quality and require additional processing steps (high pressure acid leaching (HPAL)) to produce Class 1 nickel.

Some (but not all) limonitic laterites are suitable for Class 1 nickel production.
This is not the case for saprolite laterites, nickel produced from this style of deposit feeds into the stainless steel industry as Class 2 nickel.
Where to find nickel ore?
Economic concentrations of nickel occur in nickel sulfide ores and laterite nickel ores. Rich nickel deposits are found in Australia, Indonesia, South Africa, Russia and Canada, which together account for more than 50% of global nickel resources.
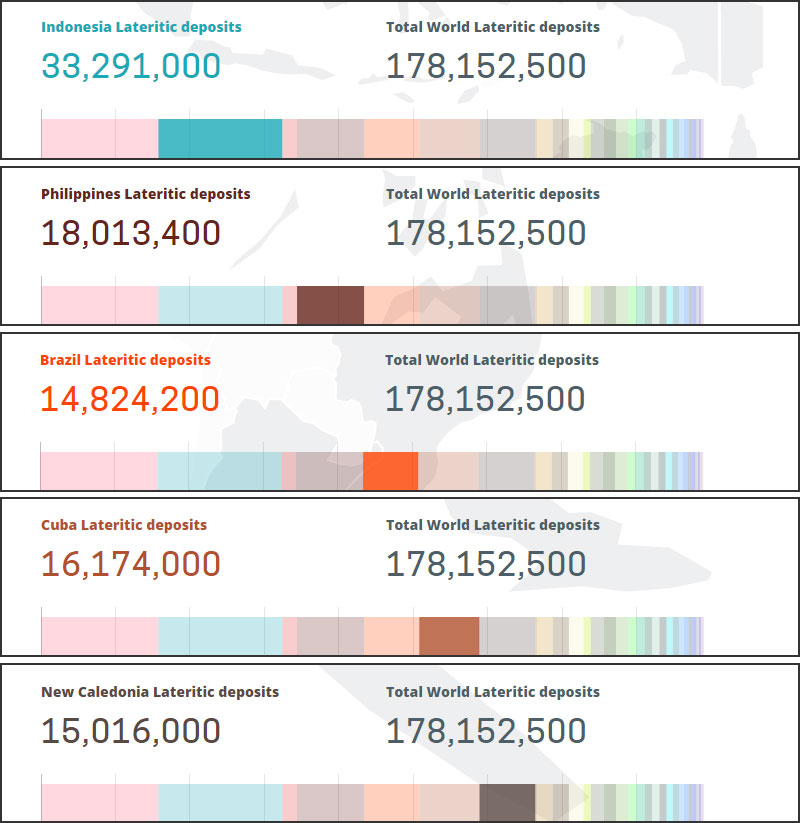
Laterite-type nickel ore (or oxide-type) resources are distributed in Indonesia, the Philippines, Brazil, Cuba and New Caledonia. These deposits and mines are mainly located in equatorial regions, and the production of such deposits has grown steadily in recent decades.
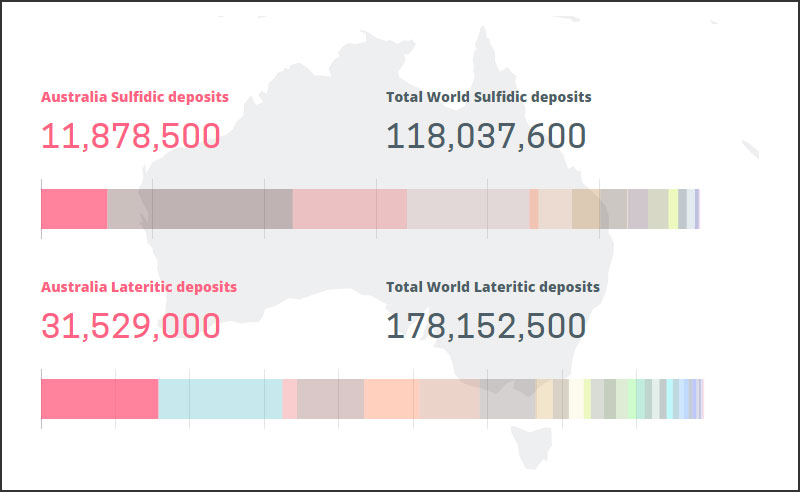
Sulfide-type nickel deposits are distributed in South Africa, Russia and Canada.

Australia has nickel sulfide deposits and laterite-type nickel deposits.
It is believed that there are additional significant nickel deposits in the sea. Technologies and proper regulatory frameworks are expected to be developed to facilitate access to these resources in the future.
Because nickel is spread across the globe, the supply chain is diverse and secure. Currently, nickel-bearing ores are mined in more than 25 countries around the world. Asia-Pacific economies account for 65% of global nickel production. Indonesia, the Philippines, Russia, Australia and Canada are the largest nickel producers.
Conclusion
Thanks to its excellent properties, nickel metal has traditionally been used to make stainless steel and is now used to make higher-performance lithium batteries. Expanded uses have led to soaring demand, which has led to increased nickel mining and processing activities.
The different types of nickel ore mentioned above go through different mining and processing procedures. To learn more, please read blog Nickel Ore Processing.