High-purity quartz sand is also called silica sand, silicon powder, and silicon dioxide powder. It is widely used in industries such as semiconductors, electronics, optical fibers, lighting, and solar energy. The soaring demand has also made people wonder how to process quartz ore into high-purity quartz sand.

Where is high purity quartz mined?
High purity quartz is a quartz product with extremely high purity of SiO2 and extremely low content of impurity after processing and purification of crystal, vein quartz, quartzite, granite pegmatite and other ores as raw materials. Economically mineable high-purity quartz deposits are rare and unevenly distributed. It is mainly distributed in 7 countries including the United States, Norway, Australia, Russia, Mauritania, China, and Canada.
The Spruce Pine deposit in USA and the Kyshtym deposit in Russia are famous quartz deposits.

High purity quartz sand processing process
We get quartz lumps by using open pit or dredging mining methods. To make high purity quartz sand, the raw quartz ore needs to go through the following quartz sand processing processes which can be divided into four stages: preparation process, pre-beneficiation process, beneficiation process and deep purification process.
- 1. Quartz sand preparation process
- 2. Quartz sand pre-beneficiation process
- 3. Quartz sand beneficiation process
- 4. Quartz sand deep purification process
Step 1: Crushing-sieving
The mined quartz ore is crushed into 1-20mm quartz particles by a crusher. Use a color sorter to select particles with smaller grayscale and higher purity.
Step 2: Calcination and water quenching
The quartz particles are first sent to the calcination equipment for high-temperature calcination, and then sent to cold water for rapid cooling. Calcination-water quenching can not only remove the bubbles and water lines inside the particles, but also expose the impurities between the grain boundaries on the outer surface of the quartz particles.
Step3: Grinding or fine crushing
Feed the cracked quartz sand minerals into the rod mill or double roller fine crusher to further dissociate quartz and gangue minerals.
Step 1: Washing and desliming
Use spiral classifier, trommel screen, hydrocyclone, desliming hopper and hydraulic classifier to wash and deslim the raw quartz ore. This effectively removes clay minerals.
Step 2: Scrubbing
With the help of mechanical force and abrasive peeling force between sand grains, the thin film iron, sticky and muddy impurity minerals on the surface of quartz sand can be removed and the quartz particles can be further broken. Add proper agents to the scrubbing machine to improve the efficiency of impurity removal.
Method 1: Gravity separation
Due to the difference in density between quartz ore particles and gangue mineral particles, gravity separation equipment such as spiral chutes, and shaking tables are commonly used to separate quartz ore particles. Gravity separation has a certain effect of removing impurities, but it is difficult to meet the production requirements, so it is mostly used for rough separation.
Method 2: Magnetic separation
Magnetic separation can effectively remove iron-containing impurities in quartz particles. Hematite, limonite, biotite and other minerals in quartz particles have weak magnetic properties, so a strong magnetic separator can be used. For strong magnetic minerals such as magnetite, a weak magnetic separator or a medium magnetic separator can be used.
Method 3: Flotation
Flotation can effectively remove iron impurities existing in iron minerals, biotite, garnet and other mineral particles, as well as aluminum impurities existing in feldspar. According to the different reagents used, it can be divided into flotation with fluorine and flotation without fluorine, and the latter is more widely used. Commonly used flotation equipment includes mechanical agitation flotation machines (such as SF type, JJF type, BF type) and inflatable agitation type flotation cells (KYF type, XCF type), etc.
Method 1: High-temperature chlorination
High temperatures expose impurities within the quartz crystal lattice to its surface. The metal oxide impurities undergo chlorination reaction with the incoming chlorinating agent in a high-temperature environment, thereby generating gaseous metal chloride and volatilizing. Chlorination can effectively remove impurities in the crystal lattice.
The types of chlorinating agents can be divided into solid chlorinating agents and gaseous chlorinating agents, solid chlorinating agents are generally NaCl, CaCl2, NH4Cl, gaseous chlorinating agents are generally Cl2, HCl.
Method 2: Acid leaching
Acid leaching utilizes the characteristics that quartz is insoluble in acid (except HF) and other impurity minerals can to purify quartz sand. Dilute acid has a better effect on removing Al and Fe, and more acidic concentrated sulfuric acid, aqua regia, and hydrofluoric acid are used to remove Cr and Ti. In practice, the purification effect of acid leaching with mixed acid is better.
6 quartz sand processing processes
In practice, we usually combine these methods to purify quartz sand. The following six quartz sand processing processes (also known as quartz sand beneficiation methods) are available for reference.
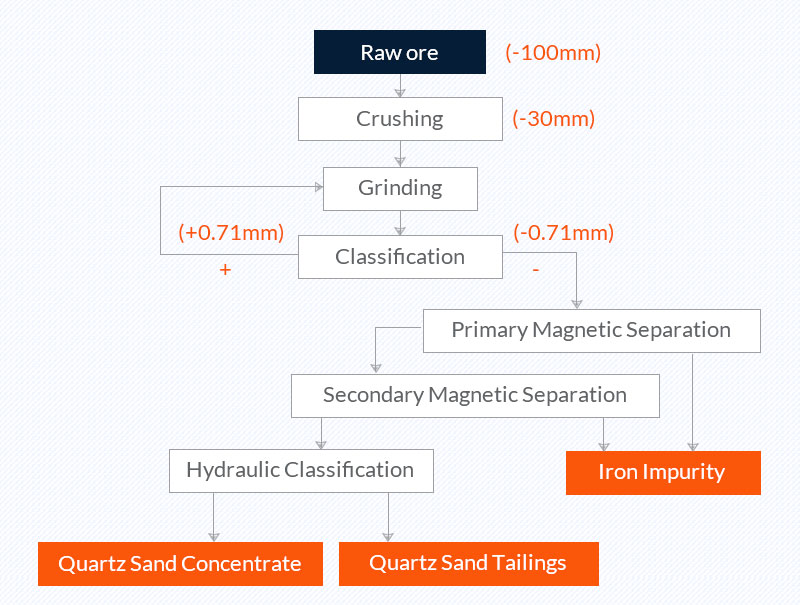
1. Comminution, classification, and magnetic separation (Quartz sand purification process of a company in Anhui)
Raw ore: quartzite from Fengyang, Anhui, with SiO2 content of 99.01% and Fe2O3 content of 0.118%;
Quartz sand concentrate: The Fe2O3 content in the concentrate is 0.02%.
Picture 1 Quartz sand purification process of a company in Anhui
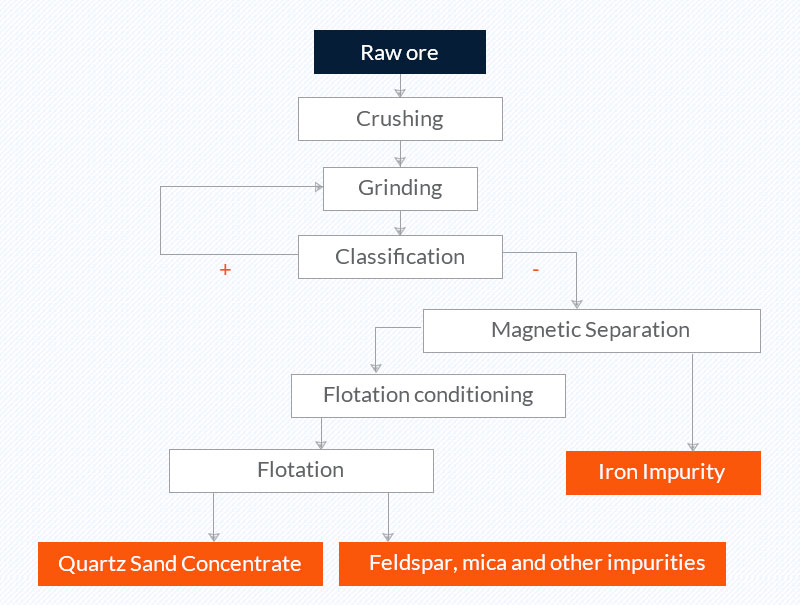
2. Comminution, classification, magnetic separation, and flotation (quartz sand purification process of a company in Fengyang, Anhui)
Raw ore: quartz sand in Anhui, its SiO2 content is 99.1%-99.3%, and the Fe2O3 content is 150-200PPM;
Quartz sand concentrate: The SiO2 content in the concentrate is 99.62%, and the Fe2O3 content is 0.0062%.
Picture 2 Quartz sand purification process of a company in Fengyang, Anhui
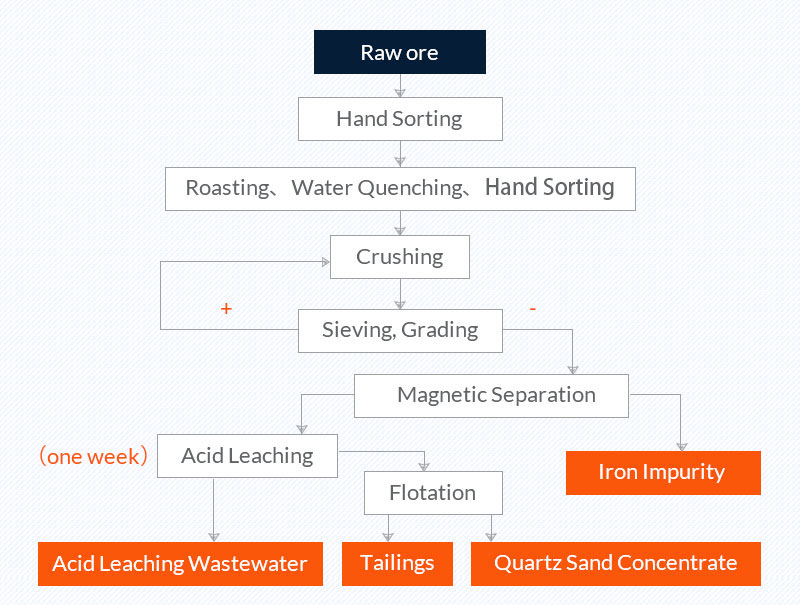
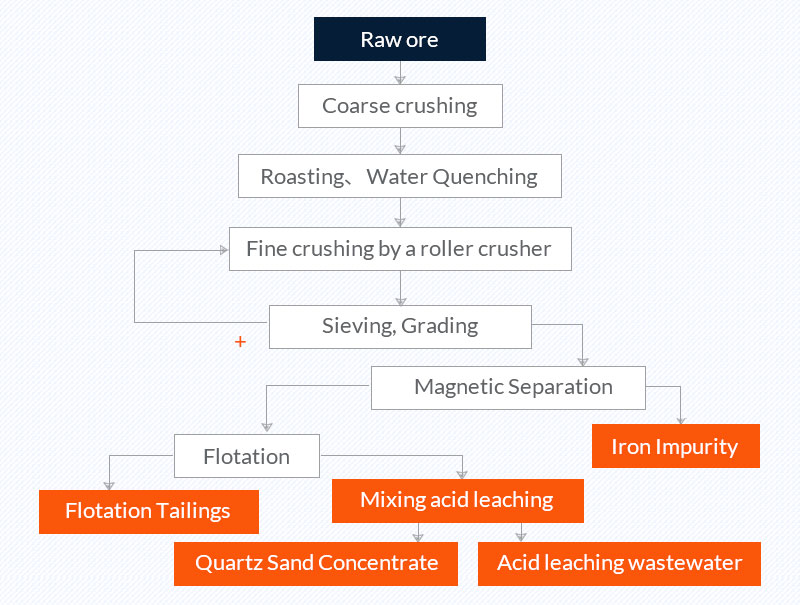
3. Roasting, water quenching, acid leaching, and flotation (quartz sand purification process of a company in Donghai, Jiangsu)
Raw ore: vein quartz with Fe content of 765.3 mg/kg.
Quartz sand concentrate: The Fe content of the concentrate is 0.35mg/kg.
Picture 3 Quartz sand beneficiation process of a company in Donghai, Jiangsu
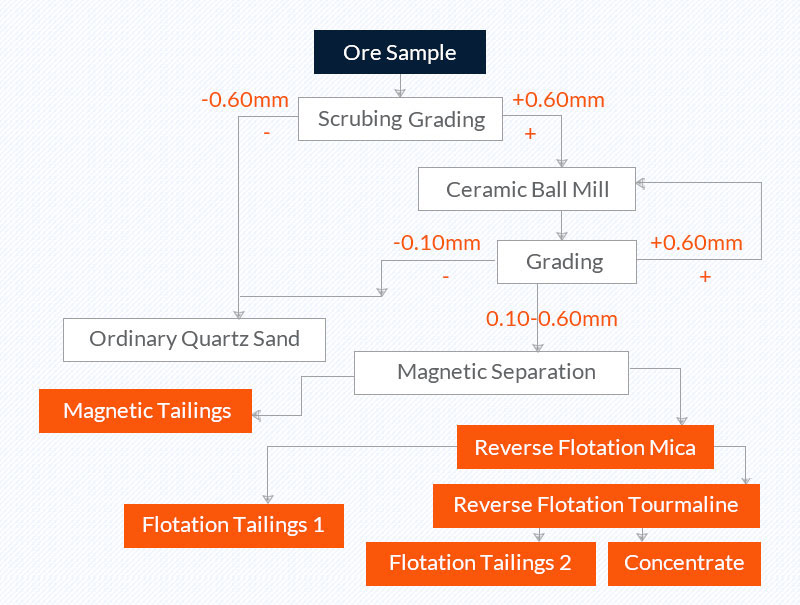
4. Scrubbing, grading, grinding, magnetic separation, reverse flotation (the purification process of kaolin associated quartz sand ore in a laboratory)
Raw ore: kaolin associated quartz sandstone in the North Sea, SiO2 content 98.32%, Fe2O3 content 1044.29μg/g;
Quartz sand concentrate: the content of SiO2 is 99.89%, and the content of Fe2O3 is 74μg/g.
Picture 4 the purification process of kaolin associated quartz sandstone in a laboratory
5. Roasting, water quenching, magnetic separation, flotation, acid leaching (quartz sandstone purification process in a laboratory)
Raw ore: quartz sandstone with SiO2 content 99.02%, Fe2O3 content 178.2μg/g;
Quartz sand concentrate: the content of SiO2 is 99.99%, and the content of Fe2O3 is 5.0μg/g.
Picture 5 quartz sandstone purification process in a laboratory
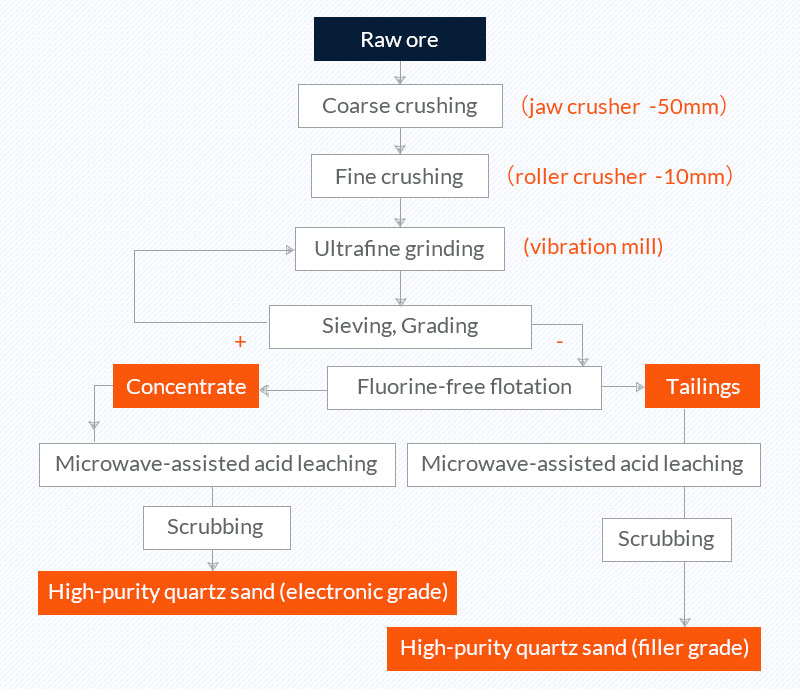
6. Ultra-fine comminution, classification, flotation, microwave-assisted acid leaching (vein quartz purification process in a laboratory)
Raw ore: vein quartz rocks from Shangrao, Jiangxi, with SiO2 content of 99.58% and Fe2O3 content of 0.03%;
Quartz sand concentrate: high-purity quartz sand (electronic grade) contains 99.98% SiO2 and 0.0003% Fe2O3; high-purity quartz sand (filler grade) contains 99.63% SiO2 and 0.0018% Fe.
Picture 6 vein quartz purification process in a laboratory
Conclusion
The high purity quartz sand (HPQ) is one of the main materials in the silica and silicon industry field.
It is produced by the crystal, quartz sandstone, and vein quartz, powder quartz, granite quartz through and by the processing and creating ways of crushing, grinding, classification, sorting, chemical washing, beneficiation leaching, thermal processing, and so on.